Biopsy Sites
Quick-Links to content:
Bullous Diseases: Skin Biopsy Sites Top
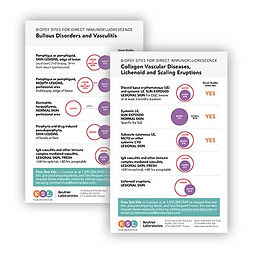
If bullous pemphigoid (BP) or epidermolysis bullosa acquisita (EBA)
is suspected, take skin biopsy with ~2/3 normal skin and ~1/3 edge
of a lesion for best DIF results.
If pemphigus is suspected, take skin biopsy with ~2/3 normal skin and
~1/3 edge of lesion plus a serum sample for the best diagnostic results.
If dermatitis herpetiformis (DH) is suspected, take normal skin ~3mm. from the edge of a lesion, as well as serum studies for IgA-EmA by indirect immunofluorescence and tTG by ELISA and eTG for best results.
If in doubt, take two DIF biopsies, one perilesional, as for pemphigoid and one normal, as for DH.
If porphyria or pseudoporphyria or lichenoid corruption or lichen planus is suspected, take skin biopsy with ~1/3 normal skin and ~2/3 edge of a lesion for best DIF results.
Bullous Diseases: Mucosal Biopsy Sites Top
If pemphigoid is suspected, take normal mucosa ~3mm. or more from a lesion or Nikolsky sign for best results.
If pemphigus or paraneoplastic pemphigus is suspected, take normal mucosa ~3mm. or more from a lesion or Nikolsky sign plus serum, for best diagnostic results.
If erosive lichen planus (LP) is suspected, take mucosal biopsy with ~2/3 normal mucosa and ~1/3 edge of a lesion, for best results.
If in doubt, take two DIF biopsies, one with edge of a lesion, as for LP, and one normal, as for pemphigoid for best results.
Connective Tissue Diseases Biopsy Sites Top
If SLE is suspected, take a biopsy of sun exposed normal skin of the forearm for DIF for LE band.
If DLE is suspected, take biopsy of a lesion in an exposed area of 3 or more month's duration for DIF and also one for light microscopy. Non-exposed areas are of little value.
If SCLE or Sjogren’s syndrome is suspected, take a sun exposed skin lesional biopsy for DIF.
If scleroderma is suspected, take biopsy of sun exposed skin for DIF and C+DIF studies. Serology studies (ANA and disease specific autoantibodies) are also indicated.
Immune Complex Vasculitis Biopsy Sites Top
If leukocytoclastic vasculitis, Henoch Schoenlein purpura or one of the other forms of immune complex vasculitis is suspected, a biopsy of a fresh lesion, less than 48 hours old, can yield a high level sensitivity. DIF studies of older lesions (more than 72 hours old) yield low sensitivity.
Hereditary Epidermolysis Bullosa (EB) Biopsy Sites Top
Preferred Skin Sites*
Normal appearing distant skin preferably upper inner arm just above the elbow.
Special Method of Obtaining Specimens
An attempt should be made prior to biopsy to induce microscopic cleavage. The only possible exception would be in patients with generalized junctional or severe generalized recessive dystrophic EB, in whom there is such inherent mechanical skin fragility as to readily demonstrate cleavage planes just with the performance of routine punch biopsy technique. In general, it is suggested that the following be done prior to biopsy of such a skin area:
-
The area to be biopsied should be sterilely prepped and then circled.
-
Using the eraser (cleaned with an alcohol swab) part of a pencil, apply firm pressure downward and then laterally in a rotary fashion (approximately 180 degrees each way for 3-5 times).
If after lifting up the pencil eraser it is obvious that the skin has markedly split, then this procedure should be re-done a bit less vigorously (to at most demonstrate a minimal puckering of the skin) in an adjacent area. However, if absolutely no change is visible after having tried to induce a blister, then one should still biopsy that area in the hope that microscopic cleavage will be noted. For example, the latter is usually the case with localized EB simplex although we can still usually demonstrate cleavage in distant skin if it is pretreated in this manner. A 4 mm. punch biopsy should then be taken of this area and the entire specimen then placed into immunofluorescence transport medium (BL, Zeus or Michel’s). Once the tissue is in this particular solution then it can be sent at room temperature to our laboratory for evaluation.
Under no circumstances should:
-
Lesional tissue be sent since frequently it contains multiple artifactual as well as true cleavage planes, thereby making diagnosis very difficult. In addition, the presence of a blister usually is associated with the release of enzymes which tend to digest the skin proteins such that we cannot satisfactorily get antibodies to react with the specific antigens.
-
Skin be obtained from the palms or soles, since the overall thickness of that tissue makes it very difficult to demonstrate skin cleavage or early blister formation.
* From the Director of the National Epidermolysis Bullosa Registry, Dr. Jo-David Fine.
References Top
-
Fine JD, Eady RAJ, Bauer EA et al. Revised classification system for inherited epidermolysis bullosa: Report of the second international consensus meeting on diagnosis and classification of epidermolysis bullosa. J Am Acad Dermatol. 2000; 42:1051-66.
-
Fine JD, McGrath J, Eady RA. Inherited epidermolysis bullosa comes into the new millennium: A revised classification system based on current knowledge of pathogenetic mechanisms and the clinical, laboratory, and epidemiologic findings of large well defined patient cohorts. J Am Acad Dermatol. 2000; 43:135-37.
-
Fine JD, Eady RAJ, Bauer EA et al. The classification of inherited epidermolysis bullosa (EB): Report of the Third International Consensus Meeting on Diagnosis and Classification of EB. J Am Acad Dermatol. 2008; 58:931-50.